ThermoDynamics
thermodynamics notes
What is Thermodynamics?
Well, thermodynamics is the effects of work, heat, flow, and energy on a system.
Examples:
Open System
Closed System
Isolated System
Thermal Energy - Kinetic energy in transit from one object to another due to temperature difference.
Temperature - the average kinetic energy of particles in an object.
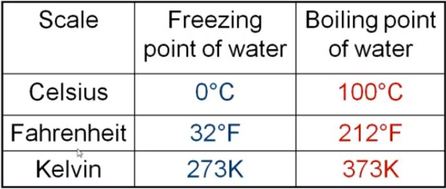
Picture below show the different ways to measure the average kinetic energy of an object.
Well, thermodynamics is the effects of work, heat, flow, and energy on a system.
Examples:
Open System
Closed System
Isolated System
Thermal Energy - Kinetic energy in transit from one object to another due to temperature difference.
Temperature - the average kinetic energy of particles in an object.
Picture below show the different ways to measure the average kinetic energy of an object.
Absolute Zero - an object that has no kinetic energy in which the molecules and atoms stop moving all together.
Thermal Equilibrium - obtained when touching objects within a system reach the same temperature
systems
Zeroth Law of Thermodynamics - If two systems are separately found to be in thermal equilibrium with a third system, the first two systems are in thermal equilibrium with each other
1st Law of Thermodynamics - thermal energy can change form and location, but it cannot be created nor destroyed
To ways thermal energy can be increased in a system:
a. preforming work in a system
b. adding thermal energy
2nd Law of Thermodynamics - Thermal energy flows from hot to cold
Entropy - the degree of disorder or uncertainty in a system
Convection - flowing cycle of hot to cold
examples: water or oil
Conduction - heat transferred by physical touch
examples: cooking pot on stove top or when someone picks up a hot plate
Thermal Equilibrium - obtained when touching objects within a system reach the same temperature
systems
Zeroth Law of Thermodynamics - If two systems are separately found to be in thermal equilibrium with a third system, the first two systems are in thermal equilibrium with each other
1st Law of Thermodynamics - thermal energy can change form and location, but it cannot be created nor destroyed
To ways thermal energy can be increased in a system:
a. preforming work in a system
b. adding thermal energy
2nd Law of Thermodynamics - Thermal energy flows from hot to cold
Entropy - the degree of disorder or uncertainty in a system
Convection - flowing cycle of hot to cold
examples: water or oil
Conduction - heat transferred by physical touch
examples: cooking pot on stove top or when someone picks up a hot plate
Conduction Equations
Convection - Q = m * c * ∆T
Rate of energy transfer - P = K * A * ∆T/L
Rate of energy transfer - P = Q/∆t
Thermal conductivity - K = P * L/A∆T
Rate of energy transfer - P = K * A * ∆T/L
Rate of energy transfer - P = Q/∆t
Thermal conductivity - K = P * L/A∆T
Variables
Q = energy transfer (joules)
m = mass of material (kilograms)
c = heat capacity (j/kg degrees C)
P = rate of energy transfer (watts)
∆t = change in time (seconds)
K = thermal conductivity
A = area of thermal conductivity (meters)
L = thickness of material (meters)
∆T = change in temperature
m = mass of material (kilograms)
c = heat capacity (j/kg degrees C)
P = rate of energy transfer (watts)
∆t = change in time (seconds)
K = thermal conductivity
A = area of thermal conductivity (meters)
L = thickness of material (meters)
∆T = change in temperature
More Notes
U-value - U = P/A * ∆T
R-value - how fast heat can escape from an object
equation: 1/U
Radiation - the process by which energy is transmitted through a medium, including empty space as electromagnetic waves
examples: heat and light
Kelvin = [(Fahrenheit - 32)5/9] + 273.15
Pnet = σAe(T24 - T14)
R-value - how fast heat can escape from an object
equation: 1/U
Radiation - the process by which energy is transmitted through a medium, including empty space as electromagnetic waves
examples: heat and light
Kelvin = [(Fahrenheit - 32)5/9] + 273.15
Pnet = σAe(T24 - T14)